
Akebi-Bashi Research Institute LLC
would like to contribute to the world with life-saving technology
- We are looking for collaborators. -
We are looking for collaborators from around the world who can implement our technology.
Time was up!! We propose you for
Time was up!! We propose you for
FREE of charge.
Hurry! Tsunamis are coming!
We develop and propose solutions to the problems we face.
We are particularly focused on developing technology that will save people's lives.
The problems we are aware of are, for example:
1. Rising sea levels due to global warming
2. The need to develop energy sources other than fossil fuels
3. Safe disposal methods of nuclear waste
Free access
-- Presentation Deck --
Free access
-- Proposal to DOE --
Free access
-- US_19360901 --
Free access
-- US_19360901_NonFinalOfficeAction --
4. Defense against infectious diseases
Free access
-- Presentation Deck 2 --
Free access
-- US_19205973 --
Below are our basic idea.
| 1. Rising sea levels due to global warming | |
|---|---|
|
This problem raises a more serious issue than leaving us with fewer places to live: the possibility of meltdowns due to submerging of nuclear power plants along our coasts. |
|
|
Sea level rises due to melting of Antarctic ice: about
60 m
by 2047(+-14 years).
→ about 120 m rise considering melting of Arctic ice and glaciers. We must hurry to waterproof our nuclear power plants and secure emergency power sources. First, let me explain why I think so. Please refer following documents: |
|
| D1 : Mora et la., “The projected timing of climate departure from recent variability”, Oct. 10, 2013, Vol. 502, nature | |
|
https://www.researchgate.net/publication/257598710_The_projected_timing_of_climate_departure_from_recent_variability "Climate departure" will be 2047, and furthermore there is poor spatial correlation (Fig. 2e) between the absolute change in the climate expected by the year 2100 (Fig. 2c) and the year at which the climate would surpass historical precedents (Fig. 2a). |
|
|
D2 Bodart et. la., "Bedmap3 updated ice bed, surface and thickness
gridded datasets for Antarctica” https://www.nature.com/articles/s41597-025-04672-y |
|
|
The sea level will rise 58m when
the ice of Antarctica melts. | |
| D3 Atkinson, "When was the last time Antarctica was ice-free?” | |
|
https://www.livescience.com/planet-earth/antarctica/when-was-the-last-time-antarctica-was-ice-free The all-melt temperature will be +8 C warmer than it is today.
So, increase in average temperature 2047 (+-14 years) Near equator: +5c or more Near poles : +8c or more
|
Next, let's look at what the state of the coastal intakes would be if such a rise in sea level were to occur. D4 Sea Level Rise Simulator https://data.earth.jaxa.jp/app/sea-level-rise-global/ I hope this gives you an idea of the magnitude of the problem we face. |
| 2. The need to develop energy sources other than fossil fuels |
|---|
|
Power sources that do not rely on fossil fuels are especially important as emergency power sources in times of disaster or at nuclear power plants. We have devised a device that extracts the alternating current found inside the Earth and converts it into practical current and voltage. 
|
| Product 1 : Power supplier |
The existence of alternating currents inside the Earth was discovered by Nikola Tesla. However, he gave up on putting it to practical use. The earth has conductive chemical element within the earth's crust. So, it can be tought that the earth has a big coil in it. There occurs induced current in its going through the magnetic field of the sun. 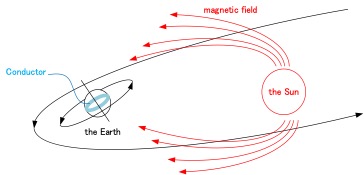  The following figure shows a concept of our device.  |
| 3. Safe disposal methods of nuclear waste |
|---|
| Summary of the proposal technic |
|
Unstable elements have an excess of neutrons in their nuclei This method allows these excess neutrons to be released extraneously. |
|
The basic idea |
|
To stabilize unstable elements, which are commonly referred to as radioactive elements, it is necessary to remove excess neutrons from the atomic nucleus. The atomic nucleus is held together by the binding energy of neutrons and protons, which is referred to as nuclear binding energy. The energy source for this binding energy is known to be the mass of the nucleons within the atomic nucleus. Therefore, this method is employed to remove neutrons from the atomic nucleus by increasing the nuclear binding energy. Specifically, a voltage is applied to the unstable element, causing protons to be drawn toward the electron orbit. The atomic nucleus converts its mass into nuclear binding energy to prevent the protons from being drawn toward the electron orbit. As described below, the influence of protons on the nuclear binding energy of protons (hereinafter referred to as EN@) is much smaller than that of electrons. Therefore, neutrons migrate outside the atom before the force relationship between protons and electrons changes. In this case, a stable atom converts the mass of its nucleons into nuclear binding energy to maintain the atomic nucleus. Now if the nuclear binding energy required is Er, the nuclear binding energy of the unstable elements is ΔE0, nuclear binding energy after a nucleus emits a neutron is ΔE1, the unstable elements emit a nucleus when following condition is satisfied. |
|
ΔE1 < ΔE0 + Er |
|
This occurs because, according to the law of entropy, the nuclei move to a state of lower potential energy. |
The nuclear binding energy |
|
Einstein’s special theory of relativity is used to express the following equation. |
|
E=mc^2 M(Z, N) = m - Δm = m - (ΔE/c^2) , where Δm is called the mass defect, and ΔE is called the nuclear binding energy. So, the following equation applies. M(Z, N) = m−Δm = m−Σ{(Zi*Ei@)} + (N*EN@) , where Ei@:Induced nuclear binding energy per electron in the i shell EN@:Induced nuclear binding energy per neutron The term +(Z*EP@) is omitted from the above equation because including this term makes it impossible to determine a definitive numerical value. This omission is considered to be proper because the induced nuclear binding energy per electron already includes the induced nuclear binding energy per proton. Calculating from known values, the following can be obtained. Δm = (Σ{(Zi*Ei@)} + (N*EN@))/c^2 (10ENe@ + 2E3S@) = 148.662 MeV/c^2 E3p@ = 11 MeV/c^2 E3d@ = 15.966 MeV/c^2 E4s@ = 28.9665 MeV/c^2 EN@ = 4.667 MeV/c^2 |
|
A constant number τ = 931 |
|
And c^2 also can be obtained as a constant number 931, that is described as τ (τ = 931). c is a velocity, so it has been impossible to obtain energy from E=mc^2. Although, it became possible to obtain energy from E=mτ. |
|
Experiment |
|
In this Experiment, the stabilization of unstable elements contained in soil through beta decay is verified. |
|
Materials (1) A soil sample 200 g of soil that had accumulated in a gutter under a railway bridge near JR Fukushima Station in Fukushima Prefecture is collected as a soil sample. (2) Detectors The left detector (Detector A) in the Fig detects the total measured amounts of β rays and γ rays. The right detector (Detector B) in the Fig detects the total measured amounts of γ ray. 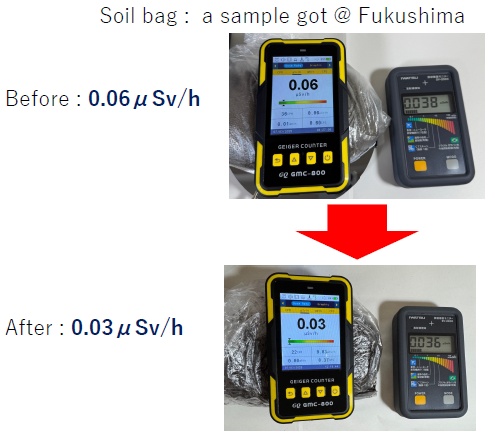 As shown in the Fig, the total amount of γ rays was approximately 0.06 μSv/h. Note that the soil in Morioka City Iwate Prefecture where this experiment was done, generally measures approximately 0.01 μSv/h. As shown in the Fig, before treatment, the total beta and gamma radiation dose minus the gamma radiation dose was approximately 0.03 μSv/h (0.06 - 0.03 = 0.03). However, as shown in the Fig, after treatment, the net beta radiation dose was nearly zero (0.03 - 0.03 = 0). The decrease in the detection values of Detector A is considered to be due to the decrease in the β ray detection values. This is thought to be caused by unstable elements contained in the soil sample undergoing β- decay and transforming into more stable elements. |
|
Conclusion Therefore, according to this method, it is possible to stabilize unstable elements. Patent applications : US19/360,901 |
|
Free access
-- Presentation Deck --
Free access
-- Proposal to DOE --
Free access
-- US_19360901 --
Free access
-- US_19360901_NonFinalOfficeAction --
|
|
4. Defense against infectious diseases |
|---|
|
Pathogens such as viruses adhere to cells at specific sites in the human body and
establish infection by entering those cells. This is because pathogens possess glycan ligands that specifically bind to specific lectins in the human body. For example, when HIV enters the human body, it first binds to DC-SIGN (dendritic cell-specific ICAM-3- grabbing non-integrin), a C-type lectin on the cell membrane surface of dendritic cells. Dendritic cells remain bound to HIV and approach T cells. HIV then presents and binds the CD4 glycan ligand possessed by the HIV envelope to the glycan receptor CD4 on the surface of the T cell and enters the T cell (see, for example, Maureen E. Taylor et al., Introduction to Glycobiology, November 1, 2005,Kagakudojin, Inc., pages 143-144). Here, even if the envelope protein changes, the lectins on the cell surface of the human body do not change. Therefore, it would require less development effort to block the lectins in the human body with glycans rather than to follow the changes in envelope proteins. We provide a glycoconjugate compound for pharmaceutical use that contains a sugar chain with three or more monosaccharides attached, which do not bind to selectins of human tissue, but binds to lectins of human tissue targeted for adhesion by an infection source.
Free access
-- Presentation Deck 2 --
Free access
-- US_19205973 --
|
 Product 5 : Glycoside compounds for pharmaceutical use Product 5 : Glycoside compounds for pharmaceutical use
|
Patent applications : US19/111,776 |
 Other Products:
Other Products:
 Other Products:
Other Products:
1. A pedal device
JP7430365B1
JP7430365B1
2. A decommissioning method and solidifying material
JP7487911B1
JP7487911B1
* Copy right licensees are now wanted.
Outline of Japanese sample of “English Grammar Acquisition Support Card
** NEW **
上記の「初学者向け英語学習キット」の著作権および特許権の使用・実施ライセンスはこちらから購入できます。
